Volume: 14 Issue: 1
Durability, Haemodynamic Performance, and Clinical Outcomes of the Carpentier Edwards PERIMOUNT Valve in the Indian Population
Year: 2026, Page: 59-68, Doi: https://doi.org/10.47799/pimr.1401.26.30
Received: March 9, 2026 Accepted: April 18, 2026 Published: May 3, 2026
Abstract
Background: The Carpentier Edwards PERIMOUNT valve is widely used for aortic valve replacement (AVR) due to its favourable haemodynamic profile and durability. This study evaluated durability, haemodynamic performance, survival, and clinical outcomes following AVR using this valve. Methods: This single-centre retrospective observational study included 42 patients who underwent AVR with the Carpentier Edwards PERIMOUNT valve between January 2016 to December 2016. Ethical approval was obtained prior to study initiation. Patients were followed at mid-term (6-months) and long-term (5-years). Clinical status was assessed using New York Heart Association (NYHA) functional class, and echocardiographic parameters included effective orifice area (EOA), peak pressure gradient (PPG), mean pressure gradient (MPG), peak systolic velocity (PSV), and left ventricular ejection fraction (LVEF). Survival analysis was performed using the Kaplan–Meier method. Results: The mean age was 67.42±6.51 years, and 76.19% of patients were male. Calcific/degenerative aortic valve disease was the most common aetiology (59.52%). Preoperatively, patients had severe valvular obstruction with a mean EOA of 1.3±0.6cm² and mean pressure gradient of 54.7±29.5mmHg. At 5-years, sustained haemodynamic improvement was observed, with EOA of 1.9±0.6cm² and persistently reduced gradients (PPG 24.0±5.0 mmHg, MPG 12.8±2.5 mmHg). Functional status improved significantly, with the majority of patients in NYHA class I–II during follow-up. Three deaths (7.14%) occurred during the follow-up period. Kaplan–Meier analysis demonstrated an estimated 5-year survival of 92.86%. Structural valve deterioration occurred in 2.38%, requiring redo AVR. Conclusions: The Carpentier Edwards PERIMOUNT valve demonstrates excellent mid-to long-term survival, sustained haemodynamic performance and significant symptomatic improvement.
Keywords: Aortic valve replacement, Carpentier Edwards PERIMOUNT valve, Effective orifice area, New York Heart Association, Peak pressure gradient
INTRODUCTION
Isolated aortic valve disease remains a significant contributor to cardiovascular morbidity and mortality worldwide, particularly among aging populations. Aortic stenosis (AS) affects approximately 2-3% of individuals aged 65 years and older, with a higher prevalence in men[1]. The aetiology of aortic valve disease is largely age-dependent and geographically variable. In patients over 70 years of age, degenerative calcific disease is the predominant cause. In contrast, among younger individuals, calcification of a congenital bicuspid aortic valve accounts for the majority of surgically treated cases. In developing countries such as India, rheumatic heart disease continues to represent an important underlying aetiology[2, 3].
Isolated aortic regurgitation (AR) constitutes the primary lesion in approximately 10–15% of patients undergoing aortic valve replacement (AVR). The pathophysiology of AR involves structural abnormalities affecting components of the functional aortic root unit, including the valve cusps, sinuses of Valsalva, and sino-tubular junction. Aetiologies range from primary valvular pathology, such as infective endocarditis or myxomatous degeneration, to aortic root disorders including connective tissue diseases and aortic dissection[4, 5].
The natural history of symptomatic severe AS is particularly dismal in the absence of surgical intervention. Historical data demonstrate a mean survival of approximately three years following the onset of angina or syncope, which declines further to 1.5–2 years once dyspnoea or heart failure develops[5, 6]. Even asymptomatic patients with severe AS carry substantial risk, with nearly one-third progressing to symptomatic status within two years and an annual risk of sudden cardiac death of 1–5%. Haemodynamic progression in moderate AS is characterized by a mean reduction in aortic valve area of approximately 0.1 cm² per year, accompanied by an average increase in mean transvalvular gradient of 7 mmHg per year and an increase in peak jet velocity of 0.3 m/s annually[7]. In contrast, chronic AR may permit prolonged compensatory left ventricular remodelling before symptom onset, whereas acute AR can precipitate rapid haemodynamic compromise[8].
Given the grave prognosis of untreated severe AS and significant AR, surgical AVR remains the definitive therapy for eligible patients. The quest for the “ideal” prosthetic valve durable, non-thrombogenic, and haemodynamically efficient has long been central to cardiac surgery. Early mechanical prostheses offered durability but were associated with thrombogenicity and lifelong anticoagulation requirements. The development of biological prostheses marked a significant advance, aiming to combine acceptable durability with superior haemodynamic and reduced thromboembolic risk[9].
Among bioprosthetic valves, bovine pericardial valves have demonstrated favourable haemodynamic characteristics due to their pliable leaflet structure and larger effective orifice area. However, structural valve deterioration (SVD), primarily driven by progressive calcification and tissue degeneration, has historically limited long-term durability[10]. Advances in tissue processing, including improved glutaraldehyde fixation techniques and anti-calcification treatments, have substantially enhanced valve longevity by preserving collagen architecture and reducing calcium deposition[11, 12].
The Carpentier Edwards Perimount valve represents a second-generation bovine pericardial bio-prosthesis designed to optimize leaflet mounting, stent flexibility, and tissue preservation, thereby improving long-term structural integrity while maintaining favourable haemodynamic. Although multiple international studies have demonstrated encouraging long-term durability and clinical performance, data from the Indian population remain relatively limited, particularly with extended follow up of five years.
The younger demographic profile, rheumatic burden, and distinct clinical characteristics of patients in India, long-term evaluation of bioprosthetic performance in this population is essential. The present study aims to assess the 5-year durability, haemodynamic performance, symptomatic improvement, and freedom from major adverse cardiac and cerebrovascular events (MACCE) following implantation of the Carpentier Edwards Perimount valve in an Indian cohort.
MATERIAL AND METHODS
This single centre, retrospective, and observational study was conducted at the Department of Cardiothoracic and Vascular Surgery from January 2023 to February 2025 after obtaining ethical approval from the Committee on Human Research Ethics, SAMSRI, no. CTVS/011/P/2023, as per the Helsinki Declaration and revised guidelines of 2013. The study population comprised 42 patients who underwent aortic valve replacement (AVR) with the Carpentier-Edwards PERIMOUNT bio-prosthesis from January 2016 to December 2016. The follow up of each patient was done at mid-term (6 months) and long-term(5-Years).
Inclusion criteria: This observational study included patients aged ≥18 years with complete medical records and documented consent for Carpentier Edwards PERIMOUNT Valve (tissue valve) implantation, managed according to the 2014 AHA/ACC guidelines. Patients undergoing isolated aortic valve replacement (AVR), as well as those receiving concomitant coronary artery bypass grafting (CABG) or other vascular procedures, were included.
Exclusion criteria: Patients aged <18 years; patients who received a mechanical valve (rather than a tissue valve); with insufficient medical record data; who underwent a Bentall were excluded.
Sample Size: Required sample size (n’) but calculated is (n)= (Zα/2+Zẞ)2 x (σd)2/Δ2
Where: n= sample size, Zα/2 = Z value for confidence level, Zẞ = Z value for power, σd= standard deviation of the paired differences and Δ = expected mean difference. 95% confidence level Zα/2 = 1.96; 80% power Zẞ = 0.84; Expected mean difference is Δ = 1.0 and Standard deviation of paired differences is σd = 2.25
n = (1.96+0.84)2 X (2.25)2 =39.70
Considering a 23% dropout rate =39.70 X 23/100 = 9.13
The required size (n’): [calculated: 39.70 +(dropout: 9.13)] = [48.83] = 49.
Assuming a 95% confidence level, 80% power, an expected mean difference in effective orifice area (EOA) of 1.0 cm², and a standard deviation of paired differences of 2.25, the required sample size was estimated to be 40 subjects with dropout 23%. The sample size was statistically validated using Open-Epi software to detect a projected increase in effective orifice area (EOA) from 1.3 preoperatively to 2.3 postoperatively. The diagnosis of aortic stenosis, aortic regurgitation, or mixed lesions (both aortic stenosis and regurgitation) was made by consulting cardiologists based on prior clinical studies and echocardiographic findings, so required sample size calculated was 49.
Preoperative Protocol: Preoperative assessment included transthoracic echocardiography (Philips IE-33, 3.5 MHz) to grade aortic pathology and coronary angiography for all patients over 45 years of age.
Anaesthetic Management: All patients received standardized general anaesthesia. Standard monitoring included electrocardiography, pulse oximetry, invasive arterial blood pressure, central venous pressure, temperature monitoring, capnography, and urine output measurement. In all patients, transesophageal echocardiography (TEE) was utilized perioperatively to assess valve pathology, confirm prosthetic valve seating, and evaluate ventricular function following cardiopulmonary bypass (CPB).
Anaesthetic induction was achieved using intravenous agents tailored to haemodynamic status, followed by endotracheal intubation and controlled mechanical ventilation. Maintenance of anaesthesia was achieved with balanced anaesthesia techniques incorporating inhalational agents, opioids, and muscle relaxants. Haemodynamic stability was maintained with vasoactive agents as required. Anticoagulation during CPB was achieved using unfractionated heparin targeting an activated clotting time (ACT) >480 seconds. Protamine sulphate was administered after weaning from CPB.
Surgical Protocol: All procedures were performed via median sternotomy under moderate hypothermic or normothermic cardiopulmonary bypass (CPB). Myocardial protection was achieved using cold blood cardioplegia (St. Thomas solution) administered via the aortic root or coronary ostia and repeated at 20-minute intervals. Following excision of the native valve and annular debridement, the PERIMOUNT valve (Model 2800TFX) was implanted using 2-0 braided polyester pledgeted sutures. Valve sizing was performed using Edwards 1161 sizers to facilitate either intra-annular placement (with everting mattress sutures) or supra-annular placement (with horizontal mattress sutures), depending on anatomical requirements. In concomitant CABG cases, distal anastomoses were performed after valve excision but before valve implantation, while proximal anastomoses were completed with a side clamp after weaning from CPB.
Postoperative Management and Follow-up: All patients were routinely ventilated overnight following the procedure and extubated in the ICU, had an ICU stay of 3 days, in the absence of any complication. After the removal of mediastinal and pericardial chest drains, enoxaparin sodium6000 IU, once daily, was started routinely in all patients for five days. Warfarin or Acitrom was not given in any of the patients as all our patients were in sinus rhythm in postoperative period. Tablet aspirin 75 mg once daily was started in patients with concomitant CABG surgery.
Follow-up assessments were conducted at six months (mid-term) and annually thereafter for 5-years (long-term) for the study. The primary endpoints were mortality and Major Adverse Cardiac and Cerebrovascular Events (MACCE), defined as readmission for heart failure, thromboembolism, infective endocarditis, or structural valve deterioration (SVD). SVD was defined as a mean transvalvular gradient >40 mm Hg or an effective orifice area (EOA) <1.0 cm² for stenosis, and an effective regurgitant orifice area >0.3 cm² for regurgitation. Data were collected via outpatient clinical evaluation, ECG, echocardiography, and structured telephone interviews. Data were analysed using Statistical Package for Social Sciences (SPSS) 23.0, IBM, Armonk, NY. Categorical variables were summarized as frequencies and proportions, while continuous variables were expressed as mean ± standard deviation. Univariate and multivariate analyses were performed to evaluate the association of age, sex, body surface area, NYHA class, diagnosis, and aetiology, and coronary angiographic findings with mortality.
Preoperative covariates potentially associated with mortality were initially explored using univariate Cox proportional hazards regression analysis, and unadjusted hazard ratios (HRs)with 95% confidence intervals (CIs) were reported. Variables of clinical relevance and those with a p-value <0.10 in univariate analysis were subsequently entered into a multivariate Cox proportional hazards regression model to identify independent predictors of mortality. Pre- and postoperative echocardiographic parameters were compared using the paired Student’s t-test. In addition, the Wilcoxon signed-rank test was applied for non-normally distributed variables. Survival analysis was performed using the Kaplan–Meier method, and survival curves were compared using the log-rank test. Hazard ratios with corresponding 95% confidence intervals were reported. A p-value <0.05 was considered statistically significant.
RESULTS
In this study, 49 eligible patients were enrolled; however, 7 were excluded due to incomplete data, leaving 42 patients available for final analysis and follow-up, as shown in [Fig. 1].
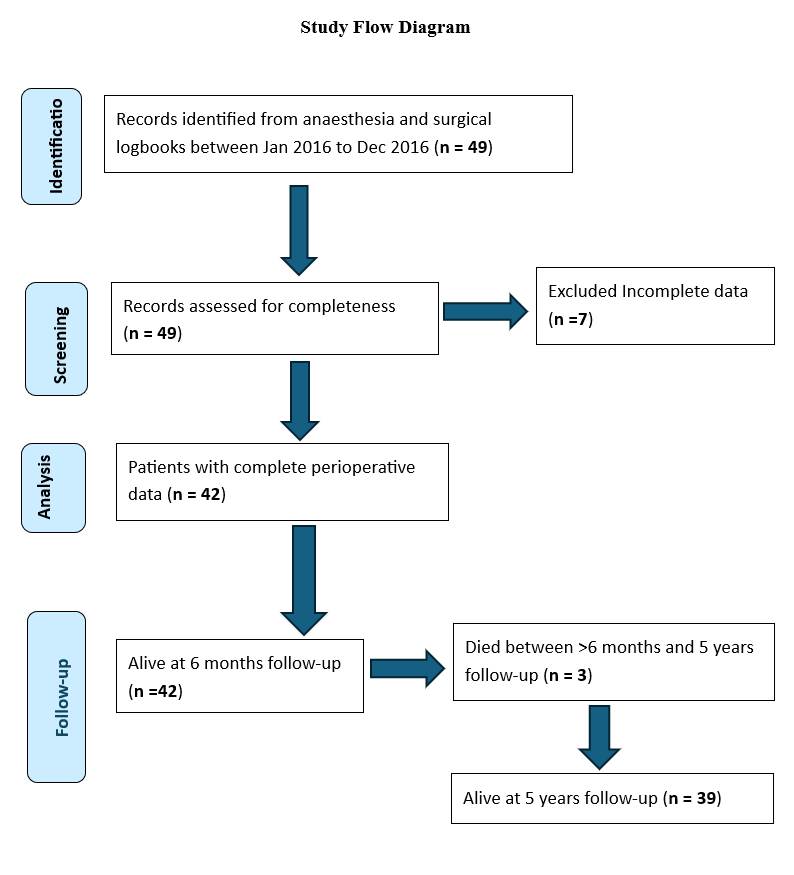
Fig. 1: Flow diagram of patient selection, inclusion, and follow-up over 5 years
In the preoperative analysis, the mean age was 67.42 ± 6.51 years, and the mean body surface area was 1.7 ± 0.1 m². The cohort was predominantly male (76.19%), with a male-to-female ratio of 3.2:1. Most patients were in New York Heart Association (NYHA) class II (57.14%), followed by class III (26.19%). Aortic stenosis was the most common diagnosis (85.72%), predominantly of calcific/degenerative origin (59.52%). Sinus rhythm was present in 95.24% of patients, and 59.52% had normal coronary angiograms, as shown in [Table. 1].
As shown in [Table. 2], isolated AVR was performed in 54.76% of patients, while 40.85% underwent AVR with CABG. Among CABG cases, most had two grafts. Redo-AVR and combined AVR with pseudoaneurysm repair were rare (2.38% each). Valve sizes of 19 mm were most frequently used (52.38%). Most cases required two doses of cardioplegia (78.57%), indicating standard operative myocardial protection.
| Variables | Number (%) (n=42) |
|---|---|
| Sex | |
| Male | 32(76.19%) |
| Female | 10(23.81%) |
| Total (M:F ratio) | 42 (3.2:1) |
| Symptomatology | |
| NYHA I | 6 (14.29%) |
| NYHA II | 24 (57.14%) |
| NYHA III | 11 (26.19%) |
| NYHA IV | 1(2.38%) |
| Diagnosis | |
| Aortic Stenosis | 36 (85.72%) |
| Aortic Regurgitation | 3 (7.14%) |
| Aortic Stenosis + Aortic Regurgitation | 3 (7.14%) |
| Aetiology | |
| Bicuspid | 10 (23.81%) |
| Calcific/Degenerative | 25 (59.52%) |
| Rheumatic | 6 (14.29%) |
| Endocarditis | 1 (2.38%) |
| ECG | |
| Sinus Rhythm | 40 (95.24%) |
| Not in Sinus Rhythm | 2 (4.76%) |
| Coronary angiogram |
|
| Normal |
25 (59.52%) |
| Single Vessel Disease |
6 (14.29%) |
| Double Vessel Disease |
8 (19.05%) |
| Triple Vessel Disease |
3 (7.14%) |
Table 1: Demographic and clinical characteristics of the study population in the pre-operative period
Data are expressed as numbers and percentage (%). ECG: Electrocardiography, NYHA: New York Heart Association
Significant haemodynamic improvement was observed postoperatively. Mean EOA increased from 1.3±0.6 to 2.0±0.5 cm², while PPG and MPG decreased markedly from 85.7±38.0 and 54.7±29.5 mmHg to 22.7±4.3 and 13.2±2.6 mmHg respectively. These improvements persisted at 6 months and 5 years, with stable gradients and preserved left ventricular function over follow-up, as shown in [Table. 3].
As shown in [Table. 4], there is significant improvement in EOA, PPG, MPG, and PSV from preoperative values to 6 months (p<0.001). LVEF showed a modest, non-significant increase. Between 6 months and 5 years, parameters remained stable with no statistically significant deterioration. MPG showed a borderline change (p=0.06), indicating preserved long-term valve haemodynamic.
As shown in [Table. 5], NYHA functional class improved significantly after surgery. At 6 months, 61.91% were in NYHA I compared to 14.29% preoperatively. At 5 years,
| Variables | Number (%) (n=42) | |
|---|---|---|
| Procedure performed |
|
|
| AVR | 23 (54.76%) | |
| AVR+CABG | 17 (40.48%) | |
| 6 | 1 Graft | |
| 8 | 2 Grafts | |
| 2 | 3 Grafts | |
| 1 | 4 Grafts | |
| Redo-AVR | 1 (2.38%) | |
| AVR +Femoral Artery Pseudoaneurysm Repair | 1 (2.38%) | |
| Size of the Valve Used | ||
| 19 MM | 22 (52.38%) | |
| 21 MM | 10 (23.81%) | |
| 23 MM | 10 (23.81%) | |
| Cardioplegia Used | ||
| Single dose | 3 (7.14%) | |
| Two doses | 33 (78.57%) | |
| Three doses | 6 (14.29%) | |
Table 2: Intra-operative data of the study population
Data are expressed as numbers and percentage (%). AVR – Aortic Valve Replacement, CABG – Coronary Artery Bypass Grafting, MM-millimetres
48.72% remained in NYHA I, with most others in class II. Postoperative complications were minimal, with 2.56% incidence stroke and tri-fascicular block case of each. One patient developed left-sided weakness during follow-up. Computed tomography (CT) of the brain revealed an acute infarct in the right frontal lobe, along with chronic infarcts involving the right posterior-temporal and occipital regions and the right cerebellar hemisphere. The patient had severe calcific aortic stenosis, and the acute infarct was most likely secondary to calcium embolism. Another patient presented with complaints of giddiness. Further evaluation revealed a tri-fascicular block on electrocardiography. The patient subsequently underwent permanent pacemaker implantation and remained asymptomatic thereafter, with no recurrence of symptoms.
As shown in [Table. 6], NYHA functional class demonstrated significant improvement following surgery. Wilcoxon signed-rank testing showed a significant reduction in NYHA class from pre-operative to 6 months (Z = −5.4, p < 0.001) and from pre-operative to 5 years (Z = −4.8, p < 0.001). There was no statistically significant difference between 6 months and 5 years (p > 0.05), indicating sustained functional benefit over time.
As shown in [Table. 7], exploratory univariable Cox regression did not identify any statistically significant associations with mortality. Age, LVEF, and MPG were not significantly associated with survival (p>0.05). This analysis is limited by the small number of events (n=3) and approximately 209 of follow-up. The wide confidence intervals reflect the imprecision of the estimates, and these findings should be interpreted with caution.
Kaplan–Meier survival analysis was performed to estimate postoperative survival following aortic valve replacement with the Carpentier Edwards PERIMOUNT valve [Fig. 2]. Of the 49 eligible patients, 7 were excluded due to incomplete data, leaving 42 patients for final analysis. Three late deaths occurred after 6 months and before 5 years of follow-up, corresponding to a mortality rate of 7.14%. The cause of death in all three patients was cardiac in origin; however, no autopsies were performed.
|
Variables |
Mean ± SD |
|---|---|
|
Preoperative echocardiography (n=42) |
|
|
Effective Orifice Area (EOA) in cm2 |
1.3 (±0.6) |
|
Peak Pressure Gradient (PPG) in mm Hg |
85.7 (±38.0) |
|
Mean Pressure Gradient (MPG) in mm Hg |
54.7 (±29.5) |
|
Peak Systolic Velocity (PSV) in m/s |
4.8 (±2.3) |
|
Left Ventricular (LV) Ejection Fraction % |
53.4 (±15.6) |
| Post-op echocardiography (n=42) |
|
| Effective Orifice Area (EOA) in cm2 |
2.0 (±0.5) |
| Peak Pressure Gradient (PPG) in mm Hg |
22.7 (±4.3) |
| Mean Pressure Gradient (MPG) in mm Hg |
13.2 (±2.6) |
| Peak Systolic Velocity (PSV) in m/s |
2.4 (±0.6) |
| Left Ventricular (LV) Ejection Fraction % |
57.6 (±7.4) |
| Echocardiography at 6 months (n=42) |
|
| Effective Orifice Area (EOA) in cm2 |
2.0 (±0.5) |
| Peak Pressure Gradient (PPG) in mm Hg |
22.0 (±4.0) |
| Mean Pressure Gradient (MPG) in mm Hg |
12.0 (±2.2) |
| Peak Systolic Velocity (PSV) in m/s |
2.5 (±0.6) |
| Left Ventricular (LV) Ejection Fraction % |
58.2 (±6.2) |
| Echocardiography at 5 years (n=39) |
|
| Effective Orifice Area (EOA) in cm2 |
1.9 (±0.6) |
| Peak Pressure Gradient (PPG) in mm Hg |
24.0(±5.0) |
| Mean Pressure Gradient (MPG) in mm Hg |
12.8 (±2.5) |
| Peak Systolic Velocity (PSV) in m/s |
2.6 (±0.6) |
| Left Ventricular (LV) Ejection Fraction % |
57.5 (±7.0) |
Table 3: Echocardiography
Data are expressed as mean ± standard deviation (SD).
| Parameter | Pre-op Mean ± SD (n=42) | 6 Months Mean ± SD (n=42) | Mean Difference | 95% CI | p-value |
|---|---|---|---|---|---|
| EOA (cm²) | 1.3 (±0.6) | 2.0 (±0.5) | +0.70 | 0.40 to 1.00 | <0.001* |
| PPG (mm Hg) | 85.7 (±38.0) | 22.0 (±4.0) | -63.7 | -75.0 to -52.0 | <0.001* |
| MPG (mm Hg) | 54.7 (±29.5) | 12.0 (±2.2) | -42.7 | -50.0 to -35.0 | <0.001* |
| PSV (m/s) | 4.8 (±2.3) | 2.5 (±0.6) | -2.3 | -2.8 to -1.8 | <0.001* |
| LVEF (%) | 53.4 (±15.6) | 58.2 (±6.2) | +4.8 | -1.5 to 7.5 | 0.12 |
| 6 Months Mean ± SD (n=42) | 5 Years Mean ± SD (n=39) | Mean Difference | 95% CI | p-value | |
| EOA (cm²) | 2.0 (±0.5) | 1.9 (±0.6) | -0.10 | -0.30 to 0.10 | 0.30 |
| PPG (mm Hg) | 22.0 (±4.0) | 24.0(±5.0) | +2.0 | -0.5 to 4.5 | 0.10 |
| MPG (mm Hg) | 12.0 (±2.2) | 12.8 (±2.5) | +0.80 | -0.05 to 1.65 | 0.06 |
| PSV (m/s) | 2.5 (±0.6) | 2.6 (±0.6) | +0.10 | -0.20 to 0.40 | 0.40 |
| LVEF (%) | 58.2 (±6.2) | 57.5 (±7.0) | -0.70 | -2.0 to 1.5 | 0.50 |
Table 4: Comparison of echocardiography using paired Student’s-t test
Data are expressed as mean ± standard deviation (SD). EOA- Effective orifice area, LVEF- left ventricular ejection fraction, MPG- mean pressure gradient, PPG- peak pressure gradient, PSV- Peak systolic velocity.
Based on available clinical records and follow-up information, these deaths were classified as presumed sudden cardiac deaths occurring during the late postoperative period. No evidence of prosthetic valve endocarditis or structural valve deterioration was documented prior to death, and none of the deaths were definitively confirmed to be valve related. The Kaplan–Meier analysis demonstrated a high overall survival probability throughout the follow-up period, with a gradual decline over time. The estimated 5-year survival was 92.9%, indicating favourable early and long-term outcomes in this cohort.
DISCUSSION
The introduction of the first-generation bovine pericardial valves was associated with structural valve deterioration (SVD) largely due to design imperfections. These early limitations prompted significant refinements in second-generation pericardial valves, such as the Carpentier-Edwards (CE) Perimount valve. Design improvements, including enhanced tissue fixation, optimized leaflet mounting, and increased stent flexibility, have preserved the intrinsic superior haemodynamic properties of pericardial tissue, while reducing the risk of early valve failure[8, 9].
Our experience with the CE Perimount valve reinforces previous reports of excellent long-term durability. In this cohort of 42 patients followed for up to 5 years, only
2.38% cases of confirmed SVD occurred, corresponding to a 97.62% freedom from SVD. This observation corroborates existing institutional data, demonstrating that modern pericardial valves provide sustained functional performance and structural integrity over extended periods[13, 14].
|
Variables |
Number (%) |
|---|---|
|
Symptomatology at 6 months (n=42) |
|
|
NYHA I |
26 (61.91%) |
|
NYHA II |
13 (30.95%) |
|
NYHA III |
3 (7.14%) |
|
NYHA IV |
0 (0%) |
|
Symptomatology at 5 years (n=39) |
|
|
NYHA I |
19 (48.72%) |
|
NYHA II |
19 (48.72%) |
|
NYHA III |
1 (2.56%) |
|
NYHA IV |
0 (0%) |
|
Postoperative complication (n=39) |
|
|
Stroke |
1 (2.56%) |
|
Tri-fascicular block |
1 (2.56%) |
Table 5: Clinical outcomes
Data are expressed as numbers and percentage (%). NYHA-New York Heart Association
| Time Point |
NYHA I |
NYHA II | NYHA III | NYHA IV | Median (IQR) | Comparison | Z (Wilcoxon) | p-value |
|---|---|---|---|---|---|---|---|---|
| Pre-operative (n=42) | 6 (14.29%) | 24 (57.14%) | 11 (26.19%) | 1 (2.38%) | Median:I (I–III) | vs 6 months | −5.4 | <0.001 |
| 6 months (n=42) | 26 (61.91%) | 13 (30.95%) | 3 (7.14%) | 0 (0%) | Median:I (I–II) | vs 5 years | −1.7 | 0.08 |
| 5 years (n=39) | 19 (48.72%) | 19 (48.72%) | 1 (2.56%) | 0 (0%) | Median:I (I–II) | vs pre-op | −4.8 | <0.001 |
Table 6: Changes in NYHA Functional Class from Pre-operative to 6 Months and 5 Years Post-Operatively
NYHA-New York Heart Association
|
Variable |
HR (95% CI) |
p-value |
|---|---|---|
|
Age |
0.95 (0.73–1.23) |
0.68 |
|
LVEF |
0.97 (0.85–1.10) |
0.62 |
|
MPG |
0.83 (0.64–1.06) |
0.14 |
Table 7: Exploratory Univariable Cox Regression Analysis of Mortality
HR-Hazard ratio, LVEF – Left Ventricular Ejection Fraction, MPG – Mean Pressure Gradient. Cox regression model: n = 42, events = 3, time at risk =209, LR χ² = 6.36, p = 0.85. Given the low number of events, the analysis is underpowered, and results should be considered exploratory.
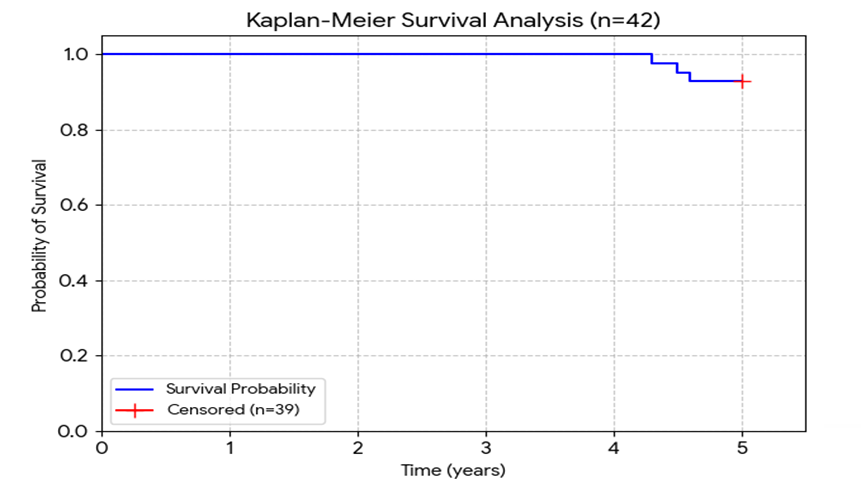
Fig. 2: Kaplan–Meier survival analysis performed to estimate patient survival following aortic valve replacement with the Carpentier Edwards PERIMOUNT valve
Valve selection in individual patients involves balancing multiple factors. Patient-specific considerations age, functional status, comorbidities, previous valve replacements, bleeding risk, and childbearing potential must be weighed against prosthesis characteristics, including haemodynamic, effective orifice area (EOA), durability, thrombogenicity, and anticoagulation requirements[9]. The primary advantage of bio-prosthetic valves is the avoidance of lifelong anticoagulation therapy, whereas the main limitation remains the potential for SVD. Despite these concerns, CE Perimount valves have demonstrated robust long-term durability, particularly in older patients. McClure et al. reported freedom from reoperation at 15 years of 34.7%, 87.4%, and 99.5% in patients aged <65, 65–75, and >75 years, respectively[15]. Similarly, Dellgren et al. [16], and Biglioli et al.[17] demonstrated favourable freedom-from-SVD rates ranging from 53% to 94% at 12–18 years, with better outcomes in patients aged ≥65 years. Bourguignon et al. analysed 2,659 patients and reported age-stratified 15- and 20-year freedom from reoperation rates of 70.8% and 38.1% (<60 years), 82.7% and 59.6% (60–70 years), and 98.1% at 15 years for older patients, highlighting the durability of CE Perimount valves across age groups[18].
Additional large-scale studies reinforce these findings. Forcillo et al. followed 2,405 patients (mean age 71 years) and reported freedom from valve reoperation of 98%, 96%, and 67% at 5, 10, and 20 years, respectively[19]. Poirer et al. reported 10- and 14-year actuarial freedom from SVD of 93% and 80%, respectively, among 598 patients[20]. These results contextualize our cohort’s outcomes[20, 21], indicating that CE Perimount valves maintain excellent structural integrity over mid- to long-term follow-up.
Valve related morbidity, including thromboembolism and bleeding, is also low with CE Perimount valves. Linearized rates for thromboembolism range from 0.3% to 2.8% per 100 patient-years, and for bleeding from 0.4% to 1.5% per 100 patient-years, according to prior studies[22-24]. In our series, no thromboembolic or major bleeding events were recorded, likely reflecting the selective use of anticoagulation only in patients with concomitant CABG and absence of preoperative atrial fibrillation in post operative period. Although underreporting or recall bias cannot be excluded, our findings corroborate other institutional experiences and emphasize the low morbidity associated with CE Perimount valves[23, 24].
Functional outcomes in our cohort were favourable. Statistically significant improvement in NYHA functional class was observed, with nearly half of patients in class I and the remainder in class II at follow-up. Echocardiographic evaluation demonstrated acceptable long-term gradients, with the highest values in patients <65 years and the lowest in those >75 years. These results support the sustained haemodynamic performance of the CE Perimount valve and the ongoing benefit of aortic valve replacement in alleviating symptomatic severe aortic valve disease[2, 9, 18].
Comparative studies between pericardial and porcine valves indicate haemodynamic superiority of pericardial tissue. Chan et al. reported lower postoperative mean and peak trans-prosthetic gradients and better survival with the Perimount valve compared to Hancock II valves[25]. Cosgrove et al. demonstrated that the CE Perimount valve provides larger effective orifice areas, lower gradients, and comparable left ventricular mass regression relative to porcine valves, supporting its favourable haemodynamic profile[26].
Recent innovations in tissue fixation, such as the Ultifix and ThermaFix methods, have the potential to further enhance the durability of pericardial valves. Ultifix uses stable amide bonding and water-soluble coupling agents to minimize calcification and cytotoxicity, while ThermaFix, zero-fixation and 3D weaving technologies aim to preserve tissue integrity for longer durations. These advances may expand the use of bioprosthetic valves to younger patients, reducing the need for anticoagulation and its associated complications[10, 11, 27].
Minimally invasive approaches for aortic valve replacement (MIAVR) [28] and transcatheter aortic valve implantation (TAVI)[29], further improve patient outcomes, reducing postoperative pain, bleeding, infection, and hospital stay. The bovine pericardial CE valves, including the Sapien valve for TAVI, have shown excellent durability and haemodynamic performance even in catheter-based procedures, broadening therapeutic options for high-risk or inoperable patients[28].
In summary, our study confirms that the Carpentier-Edwards Perimount valve provides excellent mid- to long-term durability, low rates of valve-related morbidity, and sustained functional improvement. These findings reflect previously published international data and support the use of second-generation pericardial valves as a reliable option for aortic valve replacement, particularly in patients wishing to avoid lifelong anticoagulation.
CONCLUSION
The Carpentier–Edwards Perimount valve provides satisfactory mid- to long-term outcomes in the studied population. The valve exhibited good durability with very low rates of structural valve deterioration during the follow-up period. Echocardiographic evaluation showed acceptable transvalvular gradients, with relatively higher gradients observed in younger patients and lower gradients in older individuals. Clinically, most surviving patients experienced significant symptomatic improvement, with the majority classified in New York Heart Association (NYHA) functional classes I or II. Furthermore, there was almost complete freedom from major adverse cardiac and cerebrovascular events (MACCE), supporting the safety and effectiveness of this bioprosthetic valve in aortic valve replacement.
LIMITATIONS
This study has several limitations that should be considered when interpreting the findings. First, the retrospective design inherently carries a risk of selection bias and limits the ability to establish causal relationships. Second, the relatively small sample size reduces statistical power and may limit the generalisability of the results. Third, although follow-up extended up to 5 years, this duration remains relatively short for the assessment of structural valve deterioration, which typically occurs in the longer term with bioprosthetic valves.
In addition, the predominance of smaller valve sizes, particularly 19 mm prostheses, reflects the smaller body surface area of the study population. As smaller valves are associated with higher transvalvular gradients and reduced effective orifice area, this distribution may have influenced the observed haemodynamic outcomes.
Finally, although all patients were included from the time of index surgery without exclusion of early mortality, the low number of events limits the statistical power of survival analysis. In particular, the Cox regression analysis was underpowered due to the very small number of deaths, and therefore the survival estimates should be interpreted as exploratory and hypothesis-generating rather than confirmatory.
DISCLOSURES
Conflicts of Interest: The authors have no conflicts of interest to declare.
Financing Support: This work has not received any contribution, grant or scholarship.
Confidentiality of Data: The authors declare that they have followed the protocols of their work centre on the publication of data from patients.
Protection of Human and Animal Subjects: The authors declare that the procedures followed were in accordance with the regulations of the relevant clinical research ethics committee and those of the Code of Ethics of the World Medical Association (Declaration of Helsinki).
Data availability: All data are available within the manuscript. The data sets used and analysed during the current study are available from the corresponding author on reasonable request.
References
1. Nathaniel S, Saligram S, Innasimuthu AL. Aortic stenosis: An update. World Journal of Cardiology. 2010; 2 (6). Available from: https://doi.org/10.4330/wjc.v2.i6.135
2. Tan MC, Yeo YH, San BJ, Suleiman A, Lee JZ, Chatterjee A, et al. Trends and Disparities in Valvular Heart Disease Mortality in the United States From 1999 to 2020. Journal of the American Heart Association. 2024; 13 (8). Available from: https://doi.org/10.1161/jaha.123.030895
3. Jain H, Goyal A, Khan ATMA, Khan NU, Jain J, Chopra S, et al. Insights into calcific aortic valve stenosis: a comprehensive overview of the disease and advancing treatment strategies. Annals of Medicine & Surgery. 2024; 86 (6). Available from: https://doi.org/10.1097/ms9.0000000000002106
4. Chung JC. Pathology and pathophysiology of the aortic root. Annals of Cardiothoracic Surgery. 2023; 12 (3). Available from: https://doi.org/10.21037/acs-2023-avs1-17
5. Otto CM, Nishimura RA, Bonow RO, Carabello BA, Erwin JP 3rd, Gentile F. et al. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2021; 143 (5). Available from: https://doi.org/10.1161/cir.0000000000000932
6. Dal-Bianco JP, Khandheria BK, Mookadam F, Gentile F, Sengupta PP. Management of Asymptomatic Severe Aortic Stenosis. Journal of the American College of Cardiology. 2008; 52 (16). Available from: https://doi.org/10.1016/j.jacc.2008.07.020
7. Stewart RL, Chan KL. Management of Asymptomatic Severe Aortic Stenosis. Current Cardiology Reviews. 2009; 5 (1). Available from: https://doi.org/10.2174/157340309787048103
8. Lebehn M, Vahl T, Kampaktsis P, Hahn RT. Contemporary Evaluation and Clinical Treatment Options for Aortic Regurgitation. Journal of Cardiovascular Development and Disease. 2023; 10 (9). Available from: https://doi.org/10.3390/jcdd10090364
9. Praz F, Borger MA, Lanz J, Marin-Cuartas M, Abreu A, Adamo M, et al. 2025 ESC/EACTS Guidelines for the Management of Valvular Heart Disease: Developed by the Task Force for the Management of Valvular Heart Disease of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS). European Heart Journal. 2025; 46 Available from: https://doi.org/10.1093/eurheartj/ehaf194
10. Stassen OMJA, Muylaert DEP, Bouten CVC, Hjortnaes J. Current Challenges in Translating Tissue-Engineered Heart Valves. Current Treatment Options in Cardiovascular Medicine. 2017; 19 (9). Available from: https://doi.org/10.1007/s11936-017-0566-y
11. Steitz M, Zouhair S, Khan MB, Breitenstein-Attach A, Fritsch K, Tuladhar SR, et al. A Glutaraldehyde-Free Crosslinking Method for the Treatment of Collagen-Based Biomaterials for Clinical Application. Bioengineering. 2023; 10 (11). Available from: https://doi.org/10.3390/bioengineering10111247
12. Flameng W, Hermans H, Verbeken E, Meuris B. A randomized assessment of an advanced tissue preservation technology in the juvenile sheep model. The Journal of Thoracic and Cardiovascular Surgery. 2015; 149 (1). Available from: https://doi.org/10.1016/j.jtcvs.2014.09.062
13. Kostyunin AE, Yuzhalin AE, Rezvova MA, Ovcharenko EA, Glushkova TV, Kutikhin AG. Degeneration of Bioprosthetic Heart Valves: Update 2020. Journal of the American Heart Association. 2020; 9 (19). Available from: https://doi.org/10.1161/jaha.120.018506
14. Fioretta ES, Motta SE, Lintas V, Loerakker S, Parker KK, Baaijens FPT, et al. Next-generation tissue-engineered heart valves with repair, remodelling and regeneration capacity. Nature Reviews Cardiology. 2021; 18 (2). Available from: https://doi.org/10.1038/s41569-020-0422-8
15. McClure RS, Narayanasamy N, Wiegerinck E, Lipsitz S, Maloney A, Byrne JG, et al. Late Outcomes for Aortic Valve Replacement With the Carpentier-Edwards Pericardial Bioprosthesis: Up to 17-Year Follow-Up in 1,000 Patients. The Annals of Thoracic Surgery. 2010; 89 (5). Available from: https://doi.org/10.1016/j.athoracsur.2010.01.046
16. Dellgren G, Eriksson MJ, Brodin LA, Rådegran K. Eleven years' experience with the Biocor stentless aortic bioprosthesis: clinical and hemodynamic follow-up with long-term relative survival rate. European Journal of Cardio-Thoracic Surgery. 2002; 22 (6). Available from: https://doi.org/10.1016/s1010-7940(02)00584-5
17. Biglioli P, Spampinato N, Cannata A, Musumeci A, Parolari A, Gagliardi C, et al. Long-term outcomes of the Carpentier-Edwards pericardial valve prosthesis in the aortic position: effect of patient age. J Heart Valve Dis. 2004 May;13 Suppl 1: S49-51. PMID: 15225002.
18. Bourguignon T, Bouquiaux-Stablo A, Candolfi P, Mirza A, Loardi C, May M et al. Very Long-Term Outcomes of the Carpentier-Edwards Perimount Valve in Aortic Position. The Annals of Thoracic Surgery. 2015; 99 (3). Available from: https://doi.org/10.1016/j.athoracsur.2014.09.030
19. Forcillo J, Pellerin M, Perrault LP, Cartier R, Bouchard D, Demers P, et al. Carpentier-Edwards Pericardial Valve in the Aortic Position: 25-Years Experience. The Annals of Thoracic Surgery. 2013; 96 (2). Available from: https://doi.org/10.1016/j.athoracsur.2013.03.032
20. Poirer NC, Pelletier LC, Pellerin M, Carrier M. 15-year experience with the Carpentier-Edwards pericardial bioprosthesis. The Annals of Thoracic Surgery. 1998; 66 (6 Suppl). Available from: https://doi.org/10.1016/s0003-4975(98)01110-2
21. Desai ND, Merin O, Cohen GN, Herman J, Mobilos S, Sever JY, et al. Long-Term Results of Aortic Valve Replacement With the St. Jude Toronto Stentless Porcine Valve. The Annals of Thoracic Surgery. 2004; 78 (6). Available from: https://doi.org/10.1016/j.athoracsur.2004.05.061
22. Botsile E, Mwita JC. Incidence and risk factors for thromboembolism and major bleeding in patients with mechanical heart valves: a tertiary hospital-based study in Botswana. Cardiovascular Journal of Africa. 2020; 31 (4). Available from: https://doi.org/10.5830/cvja-2020-006
23. Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin JP 3rd, Guyton RA, et al. ACC/AHA Task Force Members. 2014 AHA/ACC Guideline for the Management of Patients With Valvular Heart Disease: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014; 129 (23). Available from: https://doi.org/10.1161/cir.0000000000000029
24. Welke KF, Wu Y, Grunkemeier GL, Ahmad A, Starr A. Long-term Results after Carpentier-Edwards Pericardial Aortic Valve Implantation, with Attention to the Impact of Age. The Heart Surgery Forum. 2011; 14 (3). Available from: https://doi.org/10.1532/hsf98.20101140
25. Chan V, Kulik A, Tran A, Hendry P, Masters R, Mesana TG, et al. Long-Term Clinical and Hemodynamic Performance of the Hancock II Versus the Perimount Aortic Bioprostheses. Circulation. 2010; 122 (11_suppl_1). Available from: https://doi.org/10.1161/circulationaha.109.928085
26. Cosgrove DM. Carpentier pericardial valve. Seminars in Thoracic and Cardiovascular Surgery. 1996; 8:269 -75. PMID: 8843518.
27. Sennewald C, Pilgrim J, Aibibu D, Gereke T, Schegner P, Cherif C. Woven solutions for tissue engineering: Next-generation heart valves from fiber to function. American Heart Journal Plus: Cardiology Research and Practice. 2025; 59 Available from: https://doi.org/10.1016/j.ahjo.2025.100604
28. Doyle MP, Woldendorp K, Ng M, Vallely MP, Wilson MK, Yan TD, et al. Minimally-invasive versus transcatheter aortic valve implantation: systematic review with meta-analysis of propensity-matched studies. Journal of Thoracic Disease. 2021; 13 (3). Available from: https://doi.org/10.21037/jtd-20-2233
29. Repossini A, Fischlein T, Solinas M, DI Bacco L, Passaretti B, Grubitzsch H, et al. Stentless sutureless and transcatheter valves: a comparison of the hemodynamic performance of different prostheses concept. Minerva Cardiology and Angiology. 2018; 66 (2). Available from: https://doi.org/10.23736/s0026-4725.17.04564-9
Copyright
©2026 (Singh) et al. This is an open-access journal, and articles are distributed under the terms of the Creative Commons Attribution License CC-BY 4.0. (https://creativecommons.org/licenses/by/4.0/) which permits unrestricted use, distribution, and reproduction in any medium, provided the original authors and source are credited.
Cite this article
Singh S, Batra NA, Singh A, Baiden-Walker V, Deigheidy EM. Durability, Haemodynamic Performance, and Clinical Outcomes of the Carpentier Edwards PERIMOUNT Valve in the Indian Population. Perspectives in Medical Research 2026; 14(1):59-68 DOI: 10.47799/pimr.1401.26.30